BRAF Diagram
BRAF Inhibitors
There has recently been a paradigm shift in advanced melanoma management with the development of two new classes of drugs. The two drugs currently in clinical trials are the biggest ever development in the medical management of melanoma.
The BRAF mutation causes cells to proliferate without any control or normal programmed cell death (apoptosis). Approximately 50% of melanoma cells have the mutated BRAF gene. It is more common in the young and more likely present in metastatic disease compared with primary melanoma.
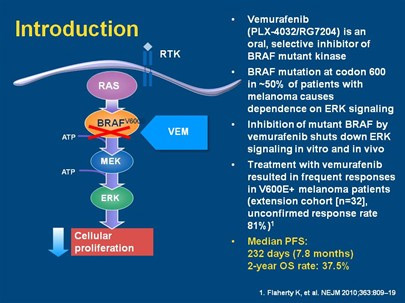
Mechanism of action
There are currently a number of companies working on a “Targeted Therapy” that blocks the mutated BRAF or MEK gene, causing cell death, shrinkage of tumours and therefore a potential for cure.
The clinical trials that have been reported show a dramatic response in patients who have the BRAF mutation. Although the follow up time is short, when compared to traditional melanoma treatment with Dacarbazine there is a 20% survival benefit at 6 months (84% vs. 64%). Overall a 63% decreased risk of death and 74% decreased risk of progression at 6 months. These statistics have continued to improve with trial data maturity.
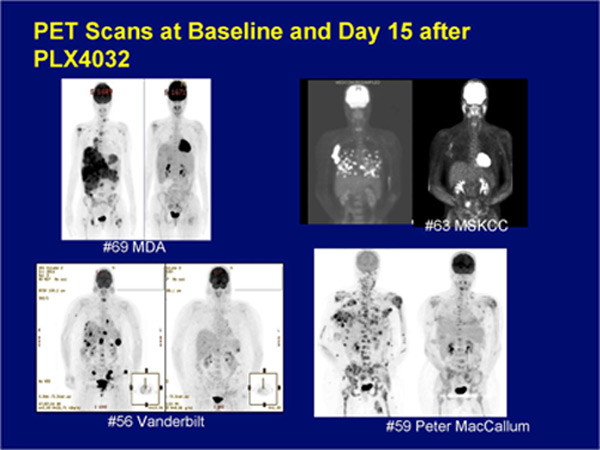
PET-CT scans showing “before” and “after” treatment with BRAF inhibitors.
The BRAF inhibitor is an oral medication taken twice a day with results seen in a few days. There are side effects as with all drugs but these are relatively minor and only 6% of patients stopped due to problems. Side effects include arthralgia, rash, fatigue, and squamous cell skin cancers.
Although this is a very exciting development in melanoma treatment it should not be considered a “magic bullet” or cure. Unfortunately resistance can develop and melanoma recurs or progresses. Currently many investigators are looking at clinical trials combining different melanoma drugs to combat resistance.
Speak to one of our Medical Oncologists about entry into a clinical trial with these drugs. The BRAF inhibitors are available off trial but cost approximately $15000 per month. Unfortunately in NZ access will be limited due to cost unless Pharmac funds treatment
